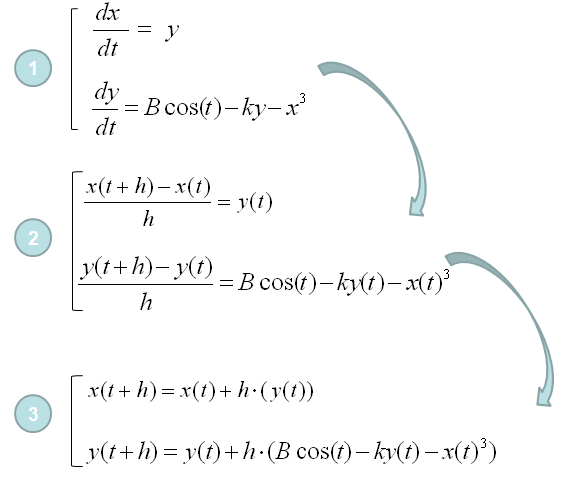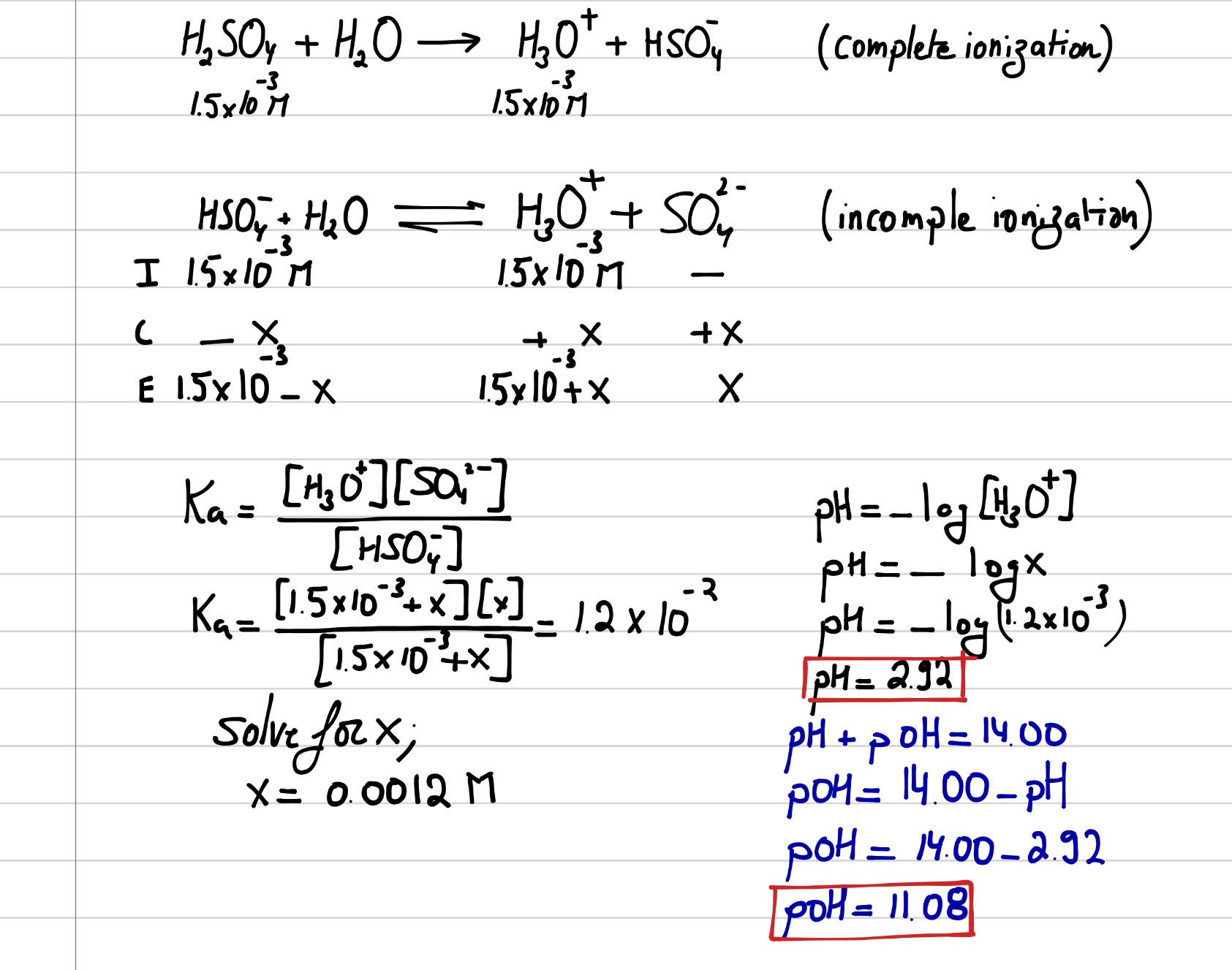equimolar mixture of A and B form an ideal solution at 300K. The vapour of this solution is condensed in second container and temperature is maintained at 300K. The vapour of second

General Solution of Trigonometric Equations:Notes on General Solution of Trigonometric Equations lUnacademyl

Explaining Why There is Only One Particular Solution of a Differential Equation Passing Through a Point, While the General Solution May Describe Infinitely Many Solutions | Calculus | Study.com
24. 200ml of an aqueous solution of a protein contains it's 1.26g.The osmotic pressure of this solution at 300K is found to be 2.57×10 3bar. The molar mass of protein will be (

Question Video: Calculating and Comparing the Concentration of Hydroxide from the Proton Concentration of an Aqueous Solution | Nagwa

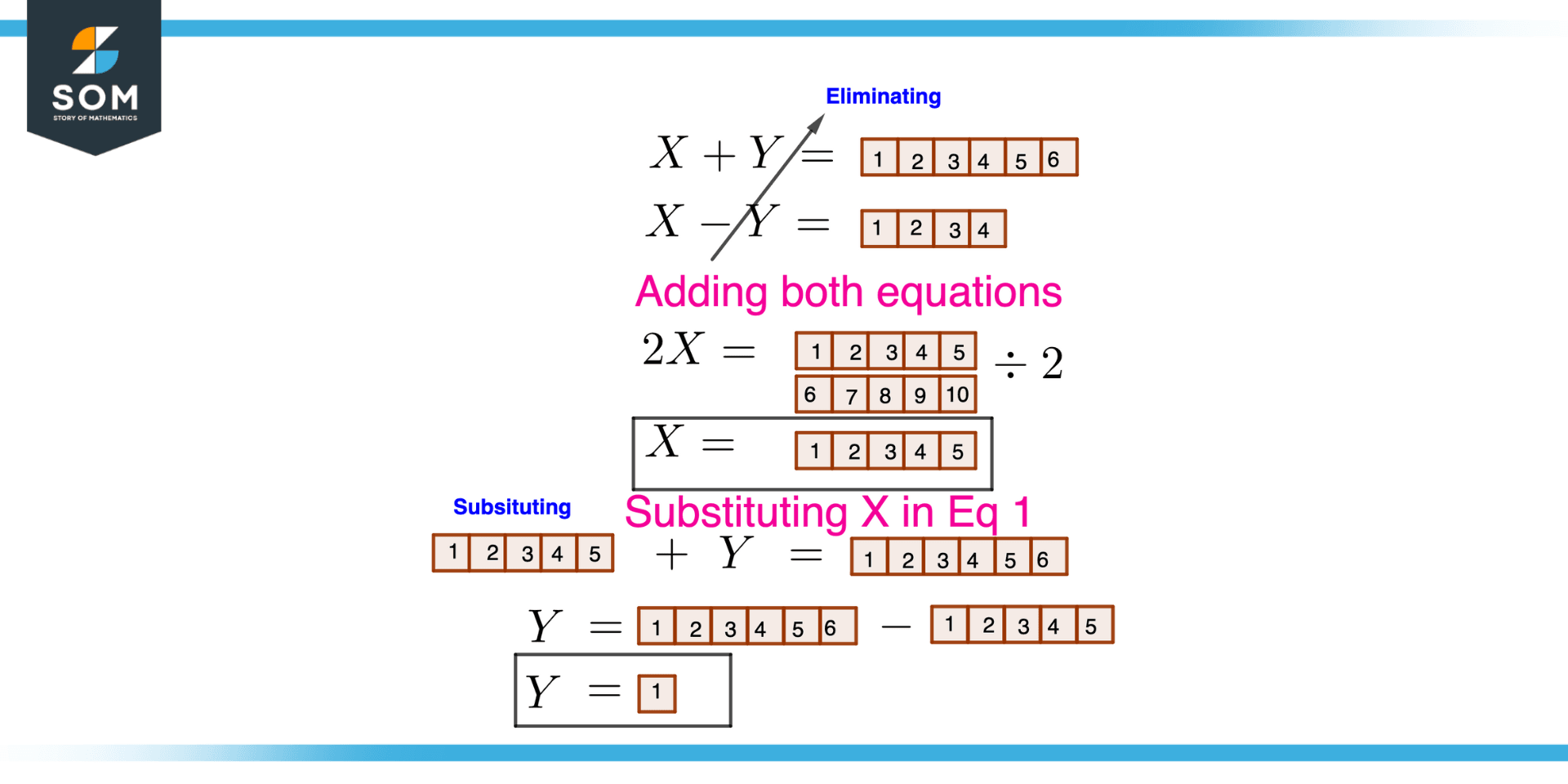
Two liquids X and Y form an ideal solution. At 300 K, vapour pressure of the solution containing 1 mol of X and 3 mol of Y is 550 mm Hg. At
equimolar mixture of A and B form an ideal solution at 300K. The vapour of this solution is condensed in second container and temperature is maintained at 300K. The vapour of second
What is the boiling point of 1 molal aqueous solution of NaCI K, 0.52 K molal (1) 99.48^° C(3) 100.52^° C(2) 98.96^° C(4) 101.04 C