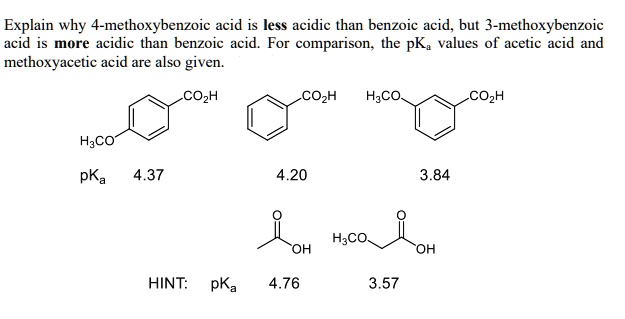
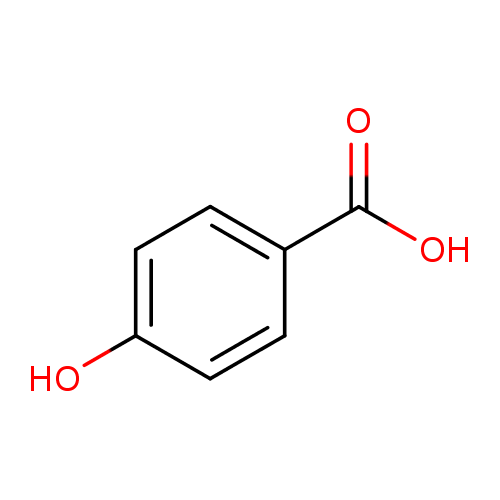
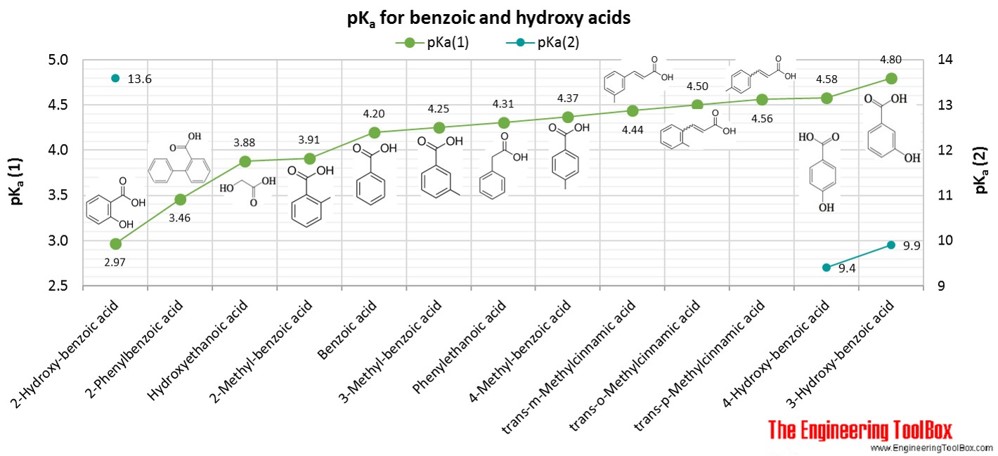
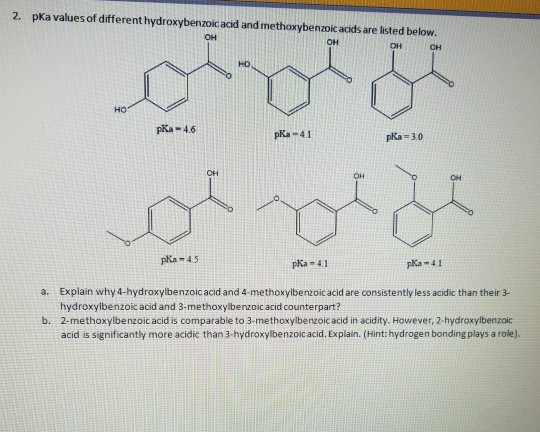
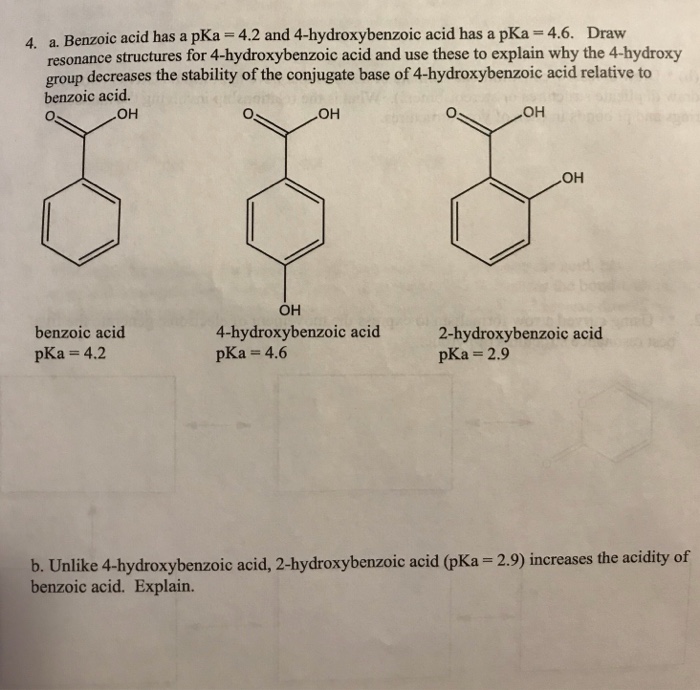
16. Arrange the following in the correct order of increasing pKa values 4 Hydroxybenzoic acid, p Anisic acid and p Toluic acid
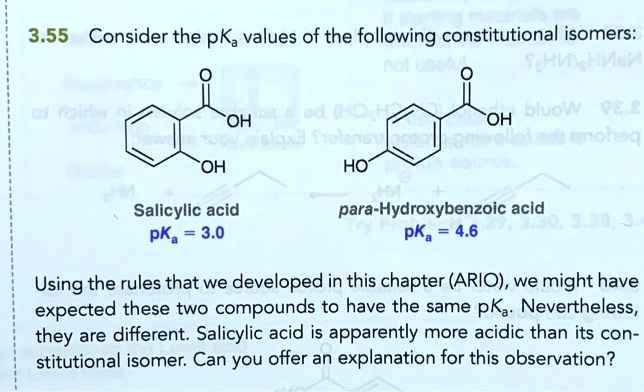
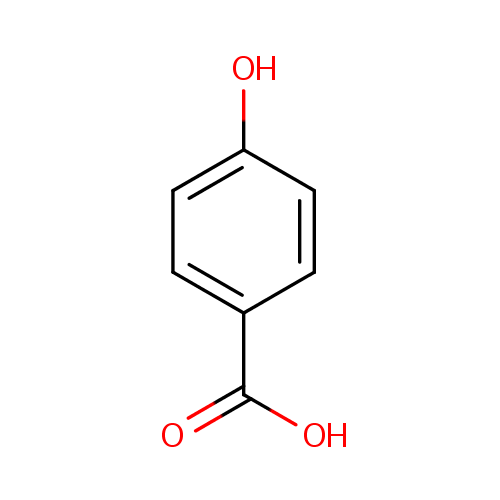
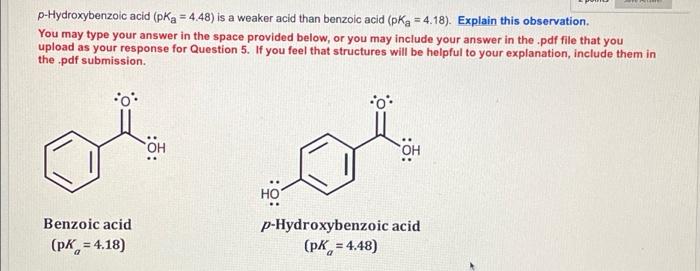
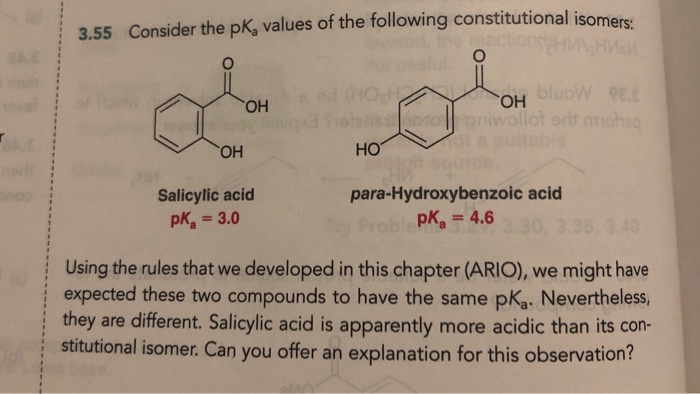
SOLVED: 3.55 Consider the pKa values of the following constitutional isomers: . ^ Uuni: ^ OH TtIn OH OH HO Salicylic acid pKa = 3.0 para-Hydroxybenzoic acid pKa 4.6 Using the rules

Deprotonation of p-Hydroxybenzoic Acid: Does Electrospray Ionization Sample Solution or Gas-Phase Structures? | Journal of the American Chemical Society

A Reliable and Efficient First Principles-Based Method for Predicting pKa Values. 2. Organic Acids | The Journal of Physical Chemistry A
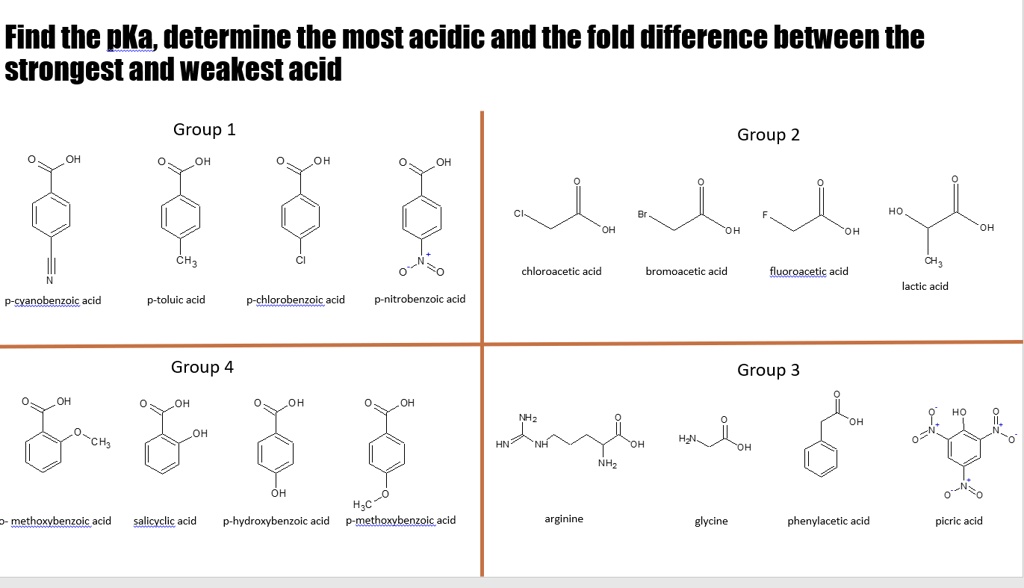
SOLVED: Find the pKa, determine the most acidic and the fold difference between the strongest and weakest acid Group 1 Group 2 CH; CH; chloroacetic acid bromoacetic acid fluoroacetic acid lactic acid

Calculated and experimental pK a values of ortho-substituted benzoic... | Download Scientific Diagram
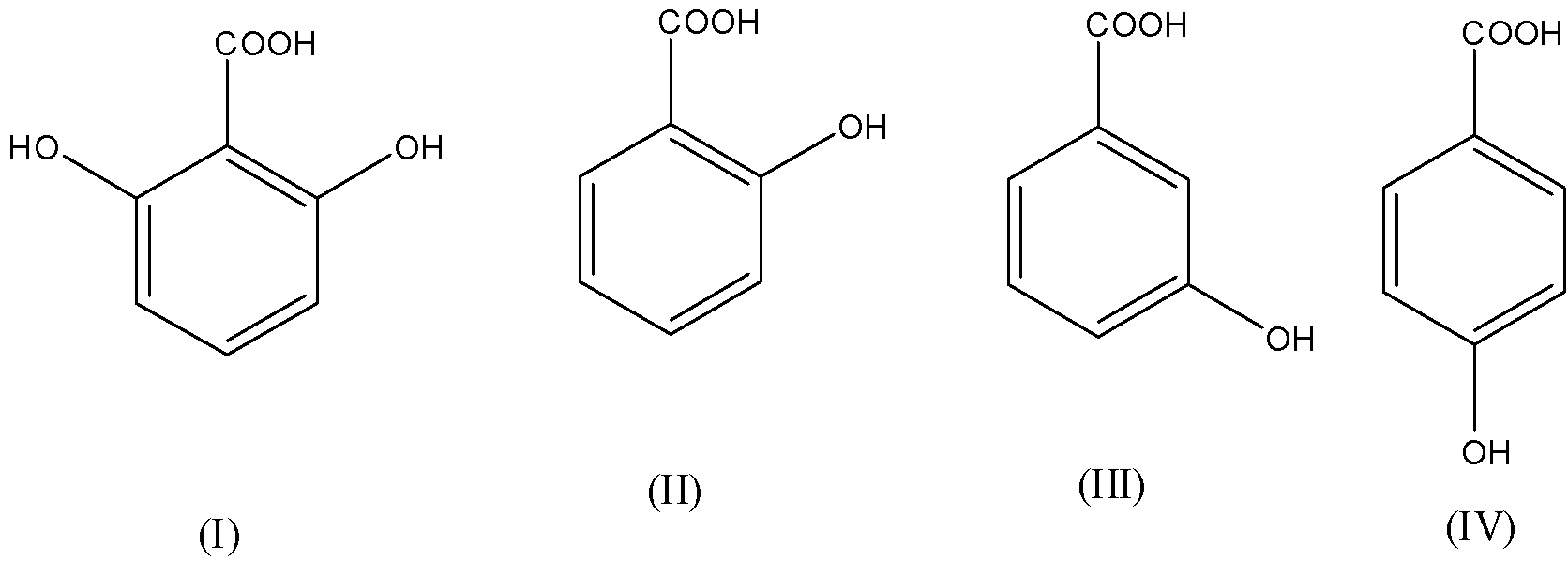
The correct order of acidity for the compounds following:\n \n \n \n \n (a)- I II III IV(b)- III I II IV(c)- III IV II I(d)- I III IV II