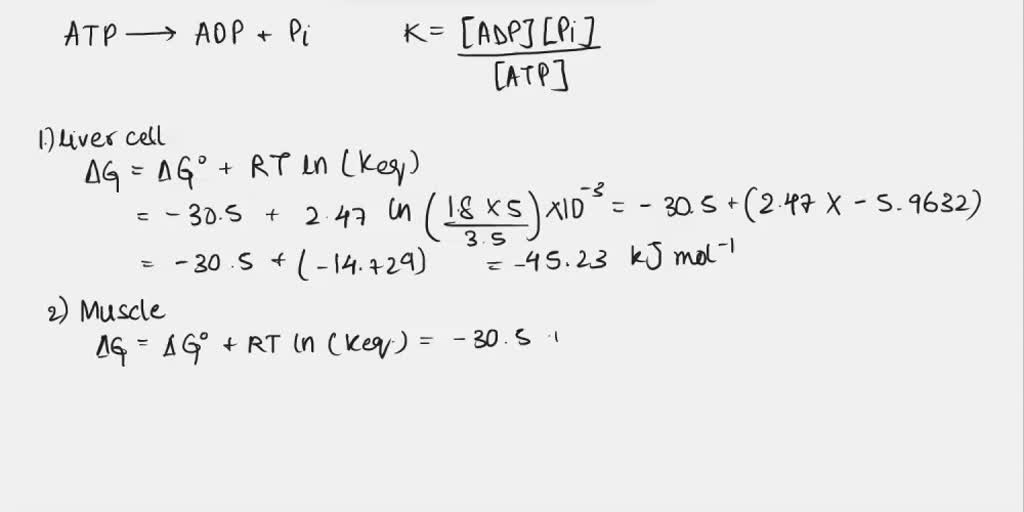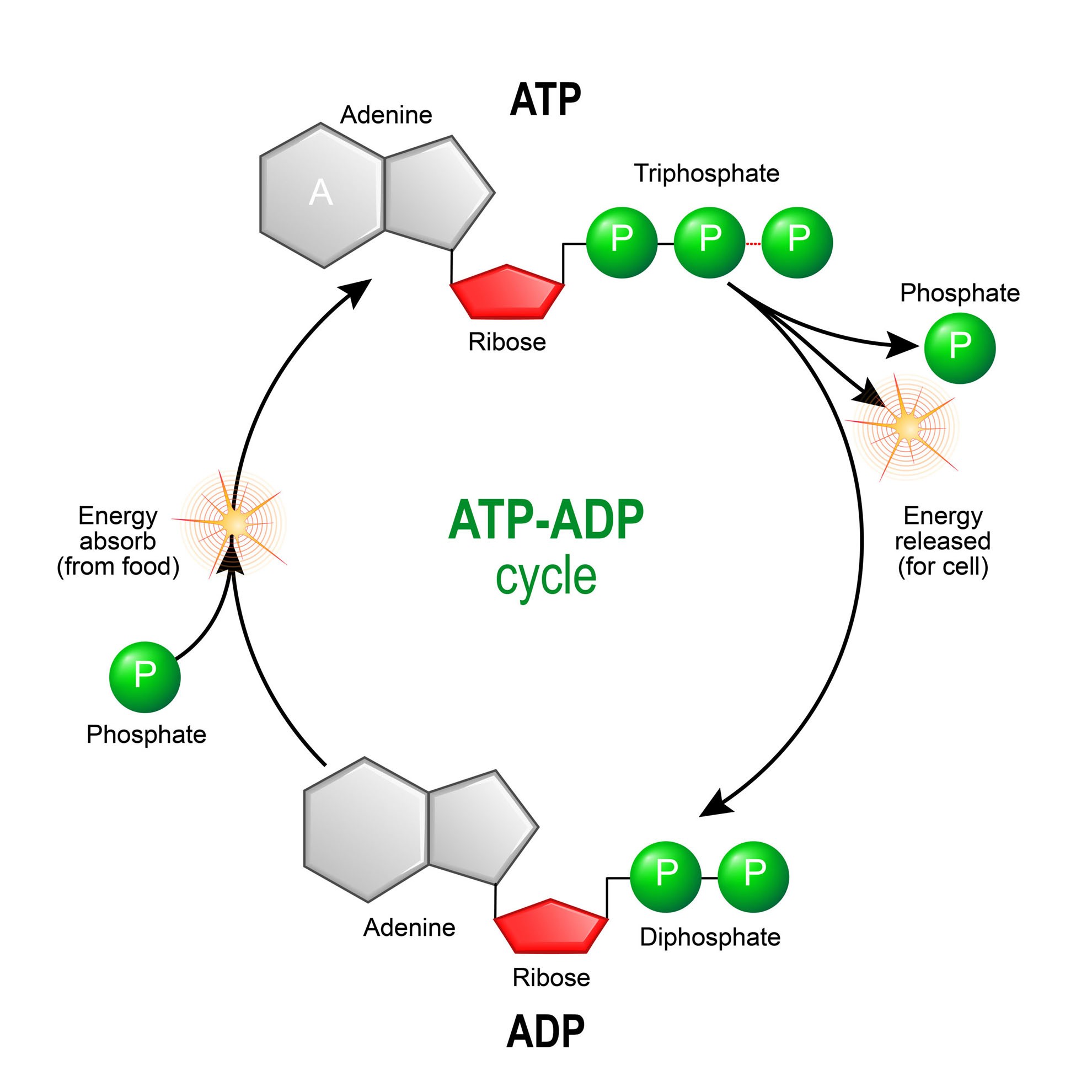
IJMS | Free Full-Text | F1·Fo ATP Synthase/ATPase: Contemporary View on Unidirectional Catalysis

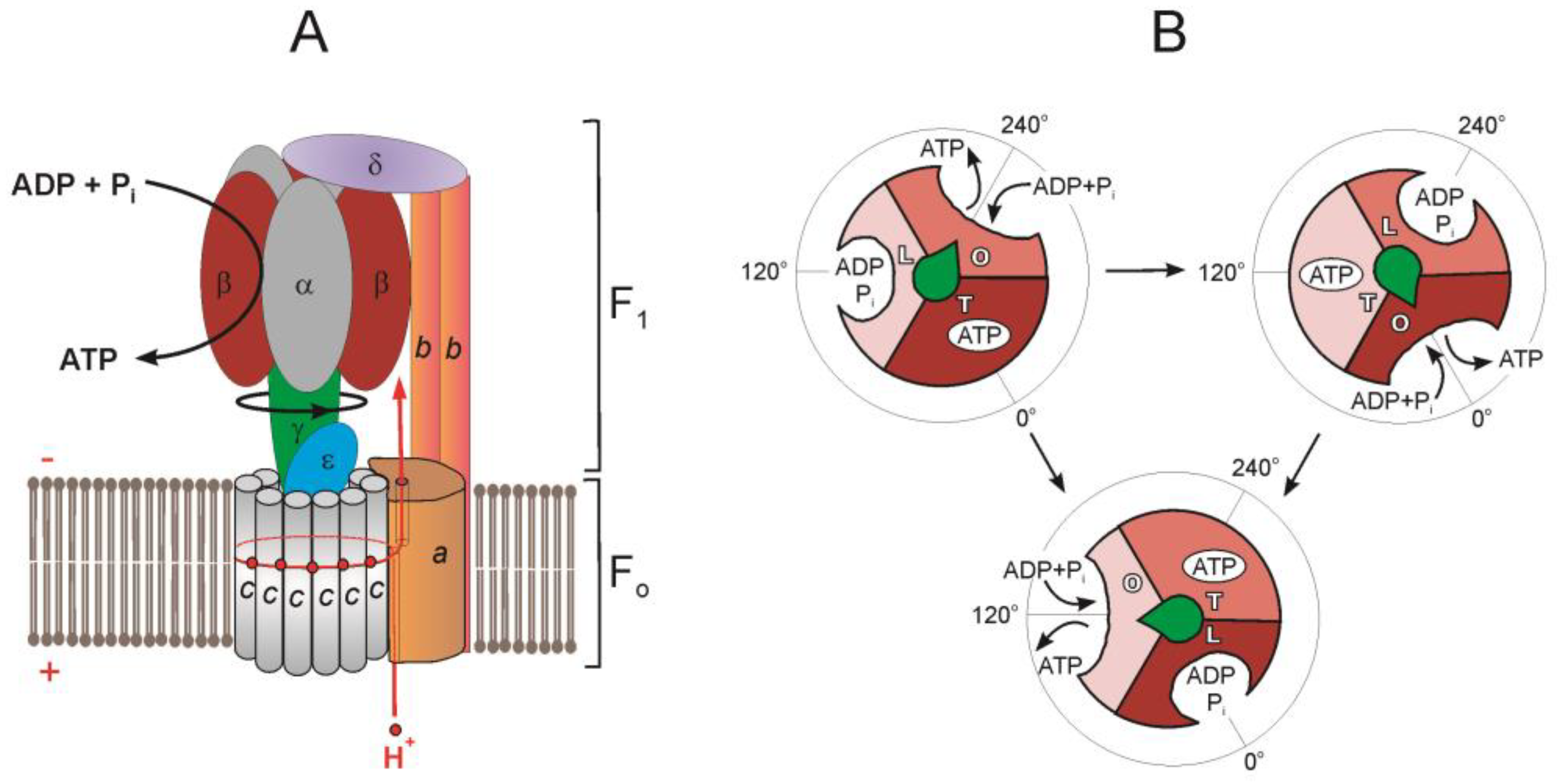
Botany - The F1 component of ATP synthase is the part that actually synthesizes ATP from ADP + Pi. Simply put, the F1 component squeezes ADP + Pi together until they form

a) The breakdown of ATP into ADP and Pi. (b) a catalyst E can enhance... | Download Scientific Diagram

Acid-labile ATP and/or ADP/Pi Binding to the Tetraprotomeric Form of Na/K-ATPase Accompanying Catalytic Phosphorylation-Dephosphorylation Cycle - ScienceDirect

Hydrolysis of ATP to ADP and inorganic phosphate (P i ) by reaction... | Download Scientific Diagram
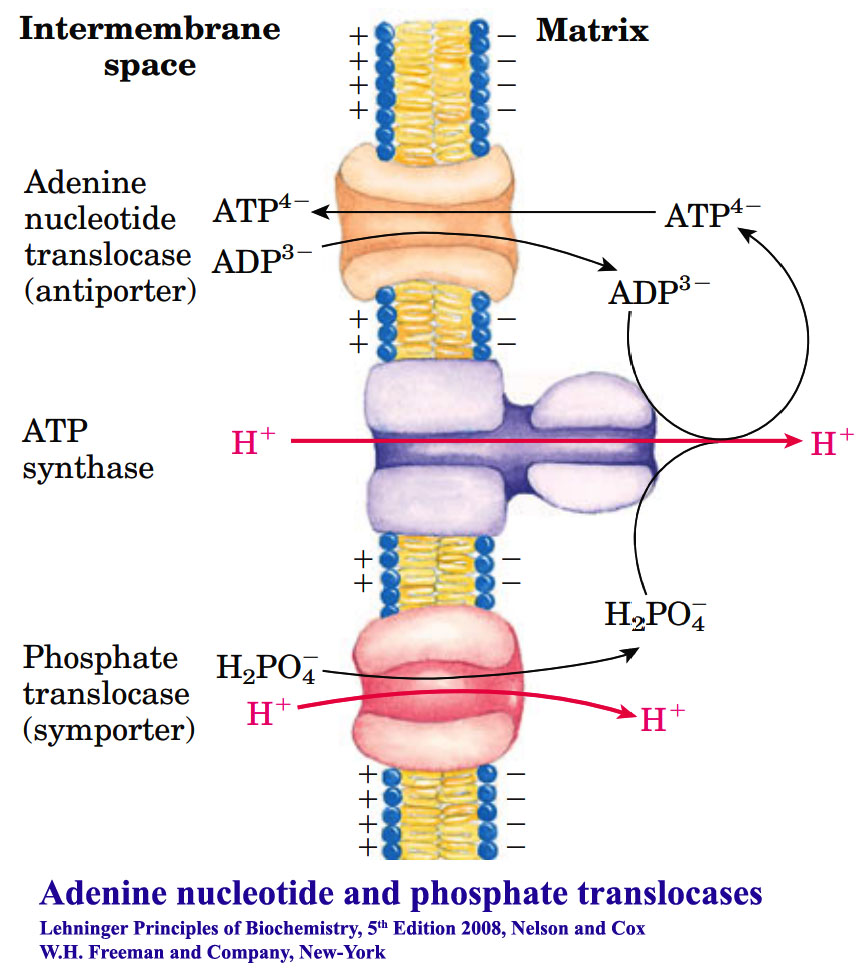
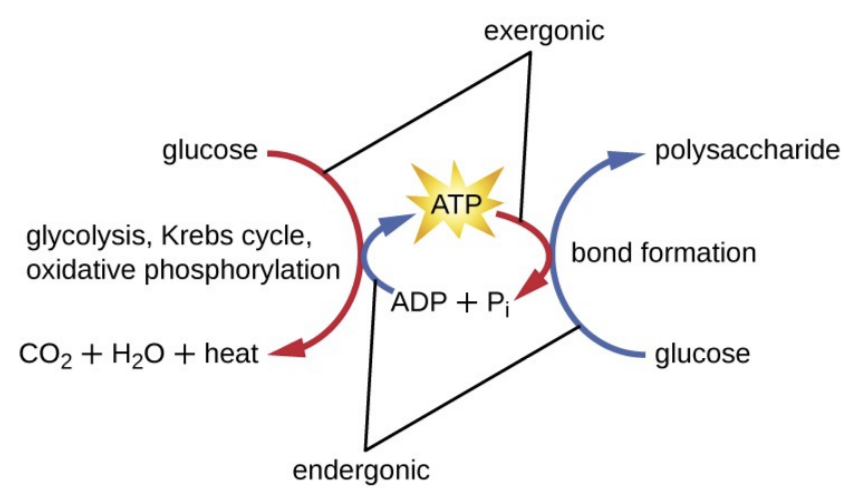
Oxidative Phosphorylation IV: Transport of ADP, Pi and cytosolic electrons to Mitochondria; Regulation of oxidative phosphorylation - Biotech MCQ

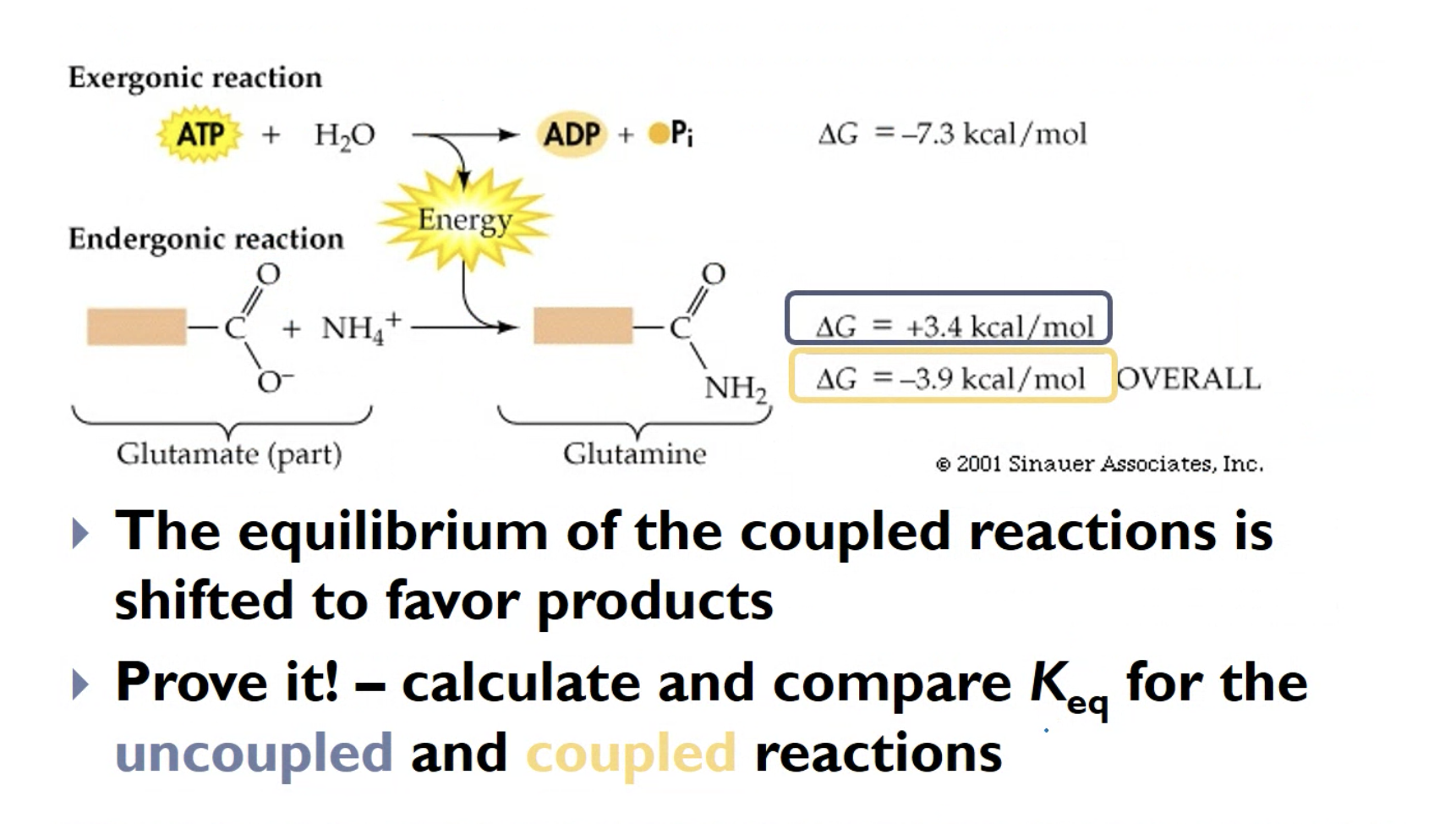
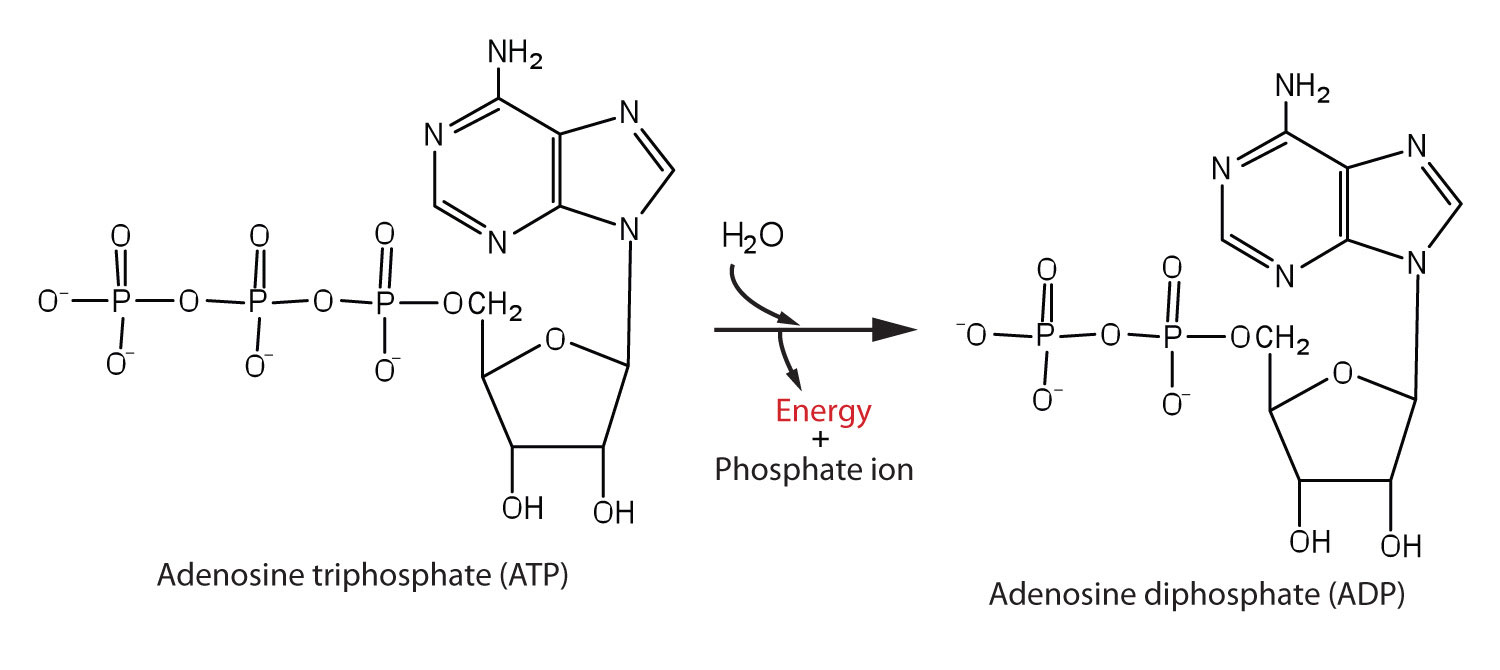
SOLVED: Question 5 Which of these equations is endergonic? Select ONE answer choice Equation 1: ATP + HzO ADP + Pi AGI =-7 kcallmol Equation 2: phosphoenolpyruvate Hzo pyruvate + Pi 4G2 =-

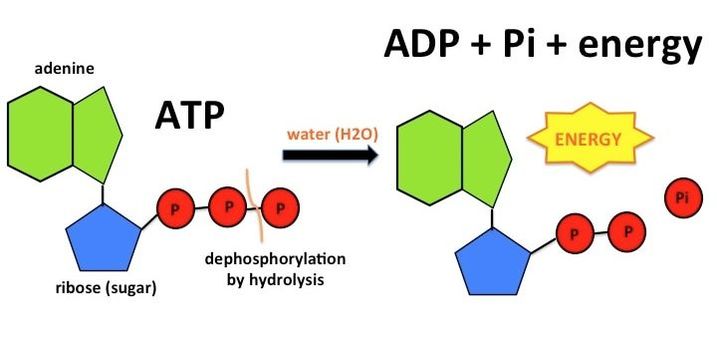
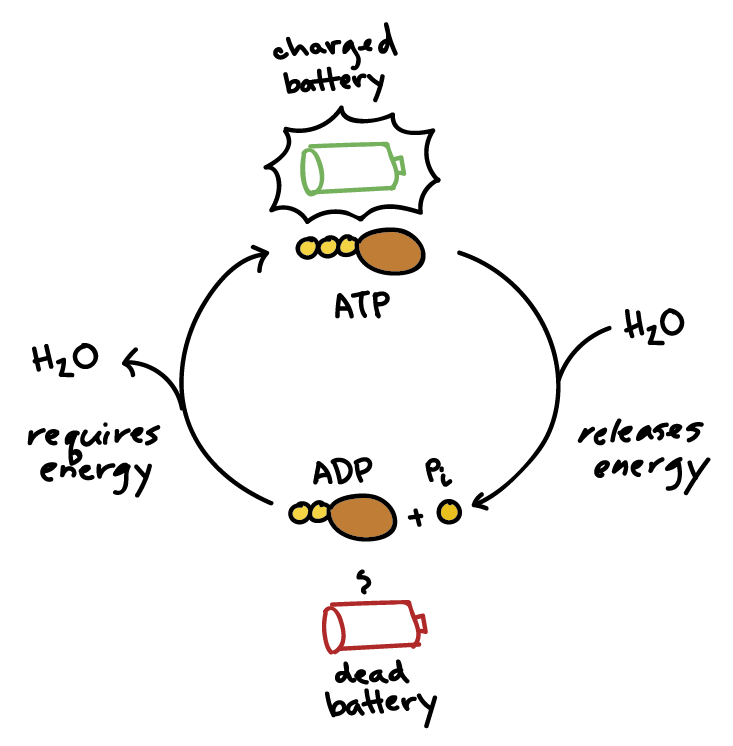
Illustrate the ATP HYDROLYSIS REACTION, its regeneration, and the delta G for both reactions. Show where energy is released in each reaction. | Homework.Study.com